A Beauty Treatment Promised to Zap Fat. For Some, It Brought Disfigurement.
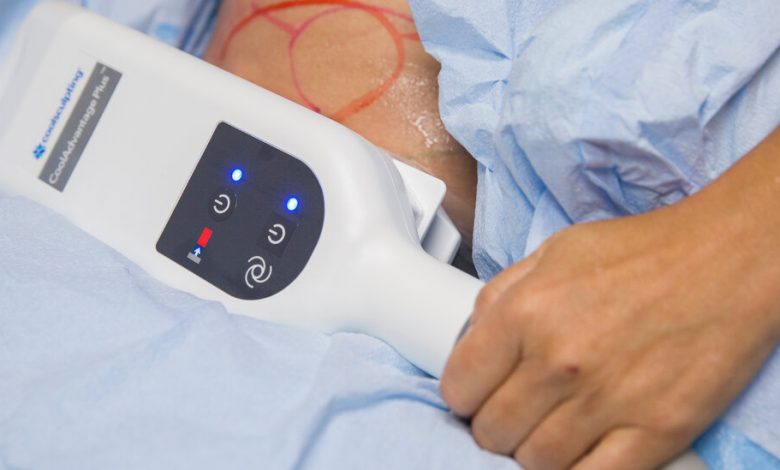
More than a dozen years ago, a medical device hit the market with a tantalizing promise: It could freeze away stubborn pockets of fat quickly, painlessly and without surgery.
The device, called CoolSculpting, was entering an already-crowded beauty industry selling flatter stomachs and tauter jaw lines, but it had an advantage: a vaunted scientific pedigree. The research behind its development came from a lab at Harvard Medical School’s primary teaching hospital, a detail noted routinely in news features and talk show segments.
The pitch worked. CoolSculpting machines are now common in dermatology and plastic surgery offices and medical spas, and the technology has generated more than $2 billion in revenue.
Cryolipolysis, the technical term for the procedure, involves placing a device onto a targeted part of the body to freeze fat cells. Patients typically undergo multiple treatments on the same area. In successful cases, the cells die and the body absorbs them.
But for some people, the procedure results in severe disfigurement. The fat can grow, harden and lodge in the body, sometimes even taking on the shape of the device’s applicator. This side effect, called paradoxical adipose hyperplasia, usually requires surgery to correct. “It increased, not decreased, my fat cells and left me permanently deformed,” the supermodel Linda Evangelista wrote in 2021 of her experience with CoolSculpting.
Allergan Aesthetics, a unit of the pharmaceutical giant AbbVie that now owns CoolSculpting, says this is rare, occurring in 0.033 percent of treatments, or about 1 in 3,000.
But a New York Times examination — drawing on internal documents, lawsuits, medical studies and interviews — indicates that the risk to patients may be considerably higher.
The company behind CoolSculpting has retained consultants who have written about low risks of P.A.H. in medical journals and online channels. It has also restricted patients from talking about the problem through confidentiality agreements and, at one point, stopped reporting the side effect to federal regulators after an auditor from the Food and Drug Administration determined that it did not qualify as a life-threatening or serious injury.
More than a dozen doctors interviewed by The Times said the manufacturer’s estimate of the risk was sharply lower than what they had observed in their practices or research — in part because the side effect can take many months to become visible, and patients don’t always connect it to CoolSculpting. Sometimes the effect is subtle, and patients believe they have just gained weight back.
“P.A.H. is likely being underreported and misdiagnosed,” a 2020 study on paradoxical adipose hyperplasia found.
In 2017, Dr. Jared Jagdeo, a dermatologist who was then a consultant for CoolSculpting’s manufacturer, and two co-authors wrote in a journal article that the side effect should be reclassified. Its increasing incidences, they wrote, met the World Health Organization’s criteria for a “common” or “frequent” adverse event, instead of a “rare” one.
Since CoolSculpting’s debut, the reported frequency of P.A.H. has quietly and steadily climbed — even in company estimates — highlighting flaws in the way the F.D.A. clears medical devices for use and monitors them after they are on the market.
The agency relies on hospitals, doctors, consumers and device manufacturers to report any “adverse events,” a system that has often been criticized as effectively turning patients into long-term test subjects. Hospitals and manufacturers are required to report deaths and serious injuries, while private doctors’ offices and consumers are not obligated to report anything.
Allergan declined to respond to detailed questions from The Times. The company emailed two statements that read, in part, “CoolSculpting has been well studied with more than 100 scientific publications.” More than 17 million treatments have been sold, Allergan noted.
The statements called the side effect rare and said it was well documented in the information the company provides for patients and doctors. Allergan also said, “We are compliant with all adverse event reporting requirements.”
Gina D’Addario, 40, who used to sell cable TV and internet services door-to-door in Syracuse, N.Y., tried CoolSculpting on her stomach in 2017. “I just wanted to pamper myself,” she said.
Ms. D’Addario said she noticed a large mass in her abdomen about nine months later. She thought it was weight gain, but dieting and exercise did not help. The bulge grew so large, she said, that her leg would bump into it when she tried to work out. It didn’t occur to her, or the many doctors she saw, that the mass could be connected to CoolSculpting, until Ms. Evangelista went public years later.
Since being diagnosed with P.A.H. in 2022, Ms. D’Addario has had multiple surgeries, including a tummy tuck and liposuction, and may need more. She said Allergan offered her $10,000 to help cover the costs, contingent on her signing a confidentiality agreement. She declined.
“I wish I loved my body back then,” she said, referring to a time before she had CoolSculpting. “To go back to that day, I wish I could, because I would never have gotten it done.”
Celebrity Cachet
The F.D.A. initially cleared CoolSculpting in 2010 for use on love handles after Zeltiq, the small company that developed the device, submitted a study of 60 subjects. That study’s modest size is typical for medical devices, while drug approvals often require much larger clinical trials. Subsequent studies led to clearances for use on other body parts.
CoolSculpting made an appearance on “Keeping Up With the Kardashians” and was praised on “The Dr. Oz Show” as a game-changing treatment that patients could get during their lunch hour. Goop, Gwyneth Paltrow’s wellness site, notes that it requires “little to no downtime.” The procedure became one of the most popular options in the body contouring industry.
The cost of CoolSculpting varies depending on the provider and the number of sessions, but on average a consumer spends $3,200, according to the manufacturer.
Part of its broad appeal is that it is not surgery. Dr. Terrence Keaney, a consultant for Allergan and a dermatologist in Arlington, Va., whose current practice has performed more than 4,000 CoolSculpting treatments since 2021, described it as the “gold standard in nonsurgical fat reduction.”
“CoolSculpting has the best risk-benefit profile,” added Dr. Keaney, who has offered the treatment for more than a decade and said he had observed two patients develop P.A.H.
But as CoolSculpting’s popularity rapidly grew, problems were quietly developing for some patients. In 2011, soon after the initial F.D.A. clearance, Zeltiq learned of a person whose treated fat had solidified into a noticeable mass, according to an internal company document obtained by The Times.
The next year, two physicians on the company’s medical advisory board — Dr. R. Rox Anderson, an inventor of CoolSculpting, and Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center — wrote an internal review of 11 patients experiencing the side effect.
Zeltiq notified the F.D.A. But it was not until 2014, more than two years after the company had learned of the side effect, that P.A.H. entered the medical literature, through an article in The Journal of the American Medical Association. Dr. Avram and Dr. Anderson were among its authors.
In an interview, Dr. Avram said he had made a concerted effort to alert the public of the side effect as soon as he learned about it from Zeltiq in 2012.
“The first thing we did was we published it out, so there could be as much awareness of it as possible,” he said.
As to the gap between the company’s findings and the article’s publication, Dr. Avram said it had taken time to analyze the data, write the report and undergo the journal’s review process. In the interim, he said, he presented information about P.A.H. at medical conferences.
Dr. Anderson did not respond to requests for comment.
A War of Numbers
When Dr. Avram and Dr. Anderson published information on the side effect in 2014, they estimated that its prevalence was 0.005 percent, or about 1 in every 20,000 treatments.
The previous year, however, a doctor advising Zeltiq had estimated the risk to be more than double that number — 0.011 percent, or about 1 in every 10,000 treatments — according to a document sent to company executives, a copy of which was obtained by The Times.
More discrepancies in data would follow, in part because the company and its consultants used the number of treatments to calculate the risk of P.A.H., while physicians observing the side effect usually used the number of patients.
For example, if two patients each underwent 10 sessions of CoolSculpting and one developed P.A.H., the company’s method would yield an incidence of 1 in 20 treatments, or 5 percent. Calculating the frequency by patient, however, would produce an incidence of 1 in 2 patients, or 50 percent.
Allergan advises getting at least two treatments, and many providers suggest more, increasing patients’ chances of ultimately developing the side effect.
Evan Mayo-Wilson, an associate professor of epidemiology at the University of North Carolina Gillings School of Global Public Health, said he thought patients would prefer to be told their overall risk, not the risk per treatment. “I think a patient wants to know, ‘What is the probability that if I start this, I’m going to have an adverse reaction?’” he said.
Dr. Jose Rodríguez-Feliz, a plastic surgeon in Miami, said he and his colleagues grew skeptical that the side effect was as rare as Zeltiq claimed.
In 20 months, four patients out of 510 who underwent CoolSculpting at their practice — about 1 in every 128 — were diagnosed with P.A.H., according to a 2016 letter to the editor of a medical journal from Dr. Rodríguez-Feliz and two co-authors.
“We felt that the difference was so big that we needed to put it out there,” Dr. Rodríguez-Feliz said in an interview.
This became a pattern. In medical journals, doctors reported observing a significantly higher incidence than what the company was reporting. In 2017, a group of doctors published that slightly more than 1 percent — or about 1 in every 100 — of their CoolSculpting patients developed the side effect. At the same time, physicians and scientists who were consultants for the manufacturer published far lower percentages.
For instance, Dr. Gordon Sasaki, a plastic surgeon who at the time consulted for Zeltiq, published a letter in response to Dr. Rodríguez-Feliz saying that the most recent incidence was 0.025 percent, or 1 in every 4,000 treatments.
Allergan, which acquired Zeltiq for $2.5 billion in 2017, now tells patients and doctors that the incidence is about 1 in every 3,000 treatments — nearly seven times the initial estimates.
The company calculates this based not on treatments performed, but on treatments sold, which can decrease the incidence it reports: Patients can buy multiple treatments in bundles and don’t necessarily use them all.
CoolSculpting has been a huge moneymaker, bringing in more than $2.2 billion between 2011 and 2019, according to company financial reports and records filed with the Securities and Exchange Commission. (Allergan, which was acquired by AbbVie in 2020, declined to share more recent sales data.)
One major beneficiary has been Massachusetts General Hospital, the Harvard-connected medical institution where the technology behind CoolSculpting was developed. In a 2011 S.E.C. filing, Zeltiq detailed a financial windfall for the hospital, including 7 percent of net sales and millions in lump sum payments tied to hitting various sales milestones.
A representative for the hospital declined to say how much money it has received from CoolSculpting.
‘That’s Not Me’
In 2015, the F.D.A. appeared concerned that Zeltiq was overlooking the risk of P.A.H., according to correspondence obtained by The Times.
The agency cautioned that a company study, examining patients up to 12 weeks after their procedures, may not have been sufficient because the fat bulges can emerge after that window of time.
The F.D.A. also noted that as of April 2013, the company had stopped reporting P.A.H. cases to the agency, even though the condition does not resolve on its own and usually requires surgery to correct. F.D.A. guidelines around “serious adverse events” state that if surgical intervention is required, or if an injury results in hospitalization or permanent bodily damage, the issue should be reported.
In this case, an F.D.A. auditor had told the company that the side effect did not meet the reporting criteria, the document said.
The Times asked the F.D.A. why its auditor had made that judgment. A spokeswoman responded that “a statement or advice given by an F.D.A. employee orally is an informal communication that represents the best judgment of that employee at that time but does not necessarily represent the formal position of the F.D.A.”
Allergan declined to respond to questions from The Times about the F.D.A. document, and the F.D.A. declined to explain what had happened after it questioned Zeltiq.
In interviews, more than a dozen dermatologists and plastic surgeons, some of whom used to offer CoolSculpting, said they believed patients were at a higher risk for developing the side effect than the company’s numbers suggest.
Dr. Erez Dayan, a plastic and reconstructive surgeon in Reno, Nev., said he had treated dozens of patients with these disfigurements. “A lot of times, they’ll feel that they caused it,” he said. “Like it’s their fault, like ‘I ate too much’ or ‘I didn’t exercise.’”
Kathryn Black, 32, a data analyst in Colorado, underwent CoolSculpting in December 2021 and then again last year for her double chin. Months later, she noticed a mass in the shape of the applicator forming in the same area. In August, she was diagnosed with P.A.H.
“The hardest part is seeing photos of myself, so I barely take any now,” she said. “When I see one, I think, ‘That’s not me.’”
Surgery to fix the growths can cost tens of thousands of dollars and leave scars.
Allergan has helped cover the cost of surgery for some patients with P.A.H., but that can be preceded by difficult negotiations. The payment is usually part of a settlement agreement that includes a confidentiality requirement, patients and doctors said.
The agreement is likely to discourage some patients from reporting their condition to the F.D.A., said Madris Kinard, a former public health analyst for the agency and the founder of Device Events, which analyzes medical device adverse event reports. Though patients can report anonymously, they may fear that it could be traced back to them, Ms. Kinard said.
Confidentiality agreements can also make patients think twice before talking about P.A.H. even with friends — let alone on social media, an important forum for sharing such information, said Dr. Rita Redberg, a cardiologist at the University of California, San Francisco, who studies the regulatory process for medical devices.
A Supermodel Sues
In 2021, Ms. Evangelista, one of the most recognizable supermodels of the 1980s and ’90s, said she had gone into a long seclusion after developing P.A.H. She sued Zeltiq and announced last summer that she had settled with the company. Ms. Evangelista declined to comment for this article.
The year she went public, the F.D.A. received over 1,100 reports of adverse events from CoolSculpting treatments — more than in the entire previous decade. Last year, the agency received more than 1,900. A majority of all the reports refer to hyperplasia.
Ms. Kinard said the spike, which she believes can be attributed in part to Ms. Evangelista, is “alarming because the device has been around for many years.”
Ms. D’Addario, who reported her condition to the F.D.A., said that before she knew what P.A.H. was, she would work out constantly, trying to lose the fat that had emerged after CoolSculpting. Now, years later, she said, she understands that it was not her fault.
But the “mental trauma” from the mysterious ways her body became deformed, and the months of not knowing what was happening, remain with her, she said: “I’m struggling now to this day. Probably worse.”
Christina Jewett and Valeriya Safronova contributed reporting.
Research was contributed by Sheelagh McNeill, Kitty Bennett, Alain Delaquérière, Kirsten Noyes and Jack Begg.




